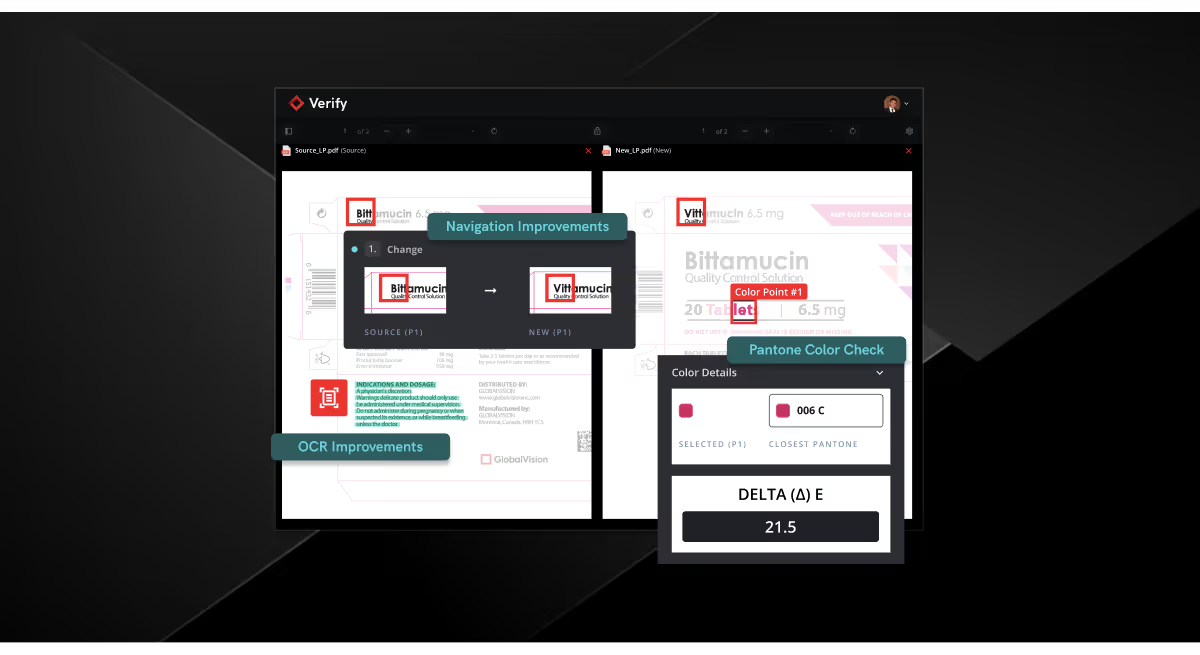
The Verify April Release: Take Control of Your Quality Inspections
Inspections should feel seamless. But when you’re dealing with complex files, tight timelines, and high-stakes content, even small inefficiencies add up fast.
Inside a Live Inline QA Demo: What Gets Caught in Seconds
The presses are rolling, and thousands of labels are flying by every minute. Suddenly, a minor defect threatens to ruin the entire run.
The Hidden Cost of Slowing the Line
For decades, plant managers assumed speed and quality were a zero-sum game, routinely slowing production to manage defects and compliance as an unavoidable cost of doing business.
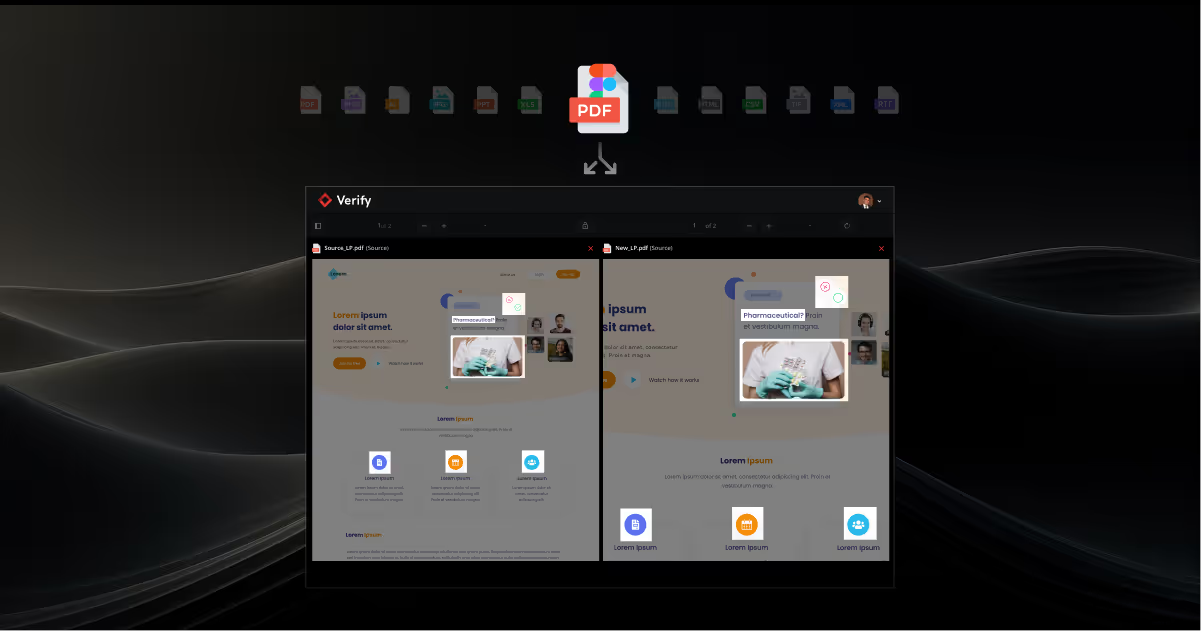
Verify CheckAI: Your New Label Compliance Assistant
Explore exactly how Verify CheckAI works, its unique features, how it protects your brand, and why its 'Human-in-the-Loop' approach is a game-changer for compliance workflows.
When Real Production Conditions Expose the Truth in QA
Line speed reveals whether your QA process is scalable, or just theoretical. But more importantly, real production conditions reveal whether your inspection system can be trusted.